
Custom Active Physical Science Curriculum
Create a customized Active Physical Science course with units from our popular Active Chemistry, Active Physics, and EarthComm programs to support the unique needs of your high school physical science program.
Available as a package or for individual purchase
Digital
On the Activate Learning Digital Platform
Kits
Materials & Supplies
Professional Learning
Three-Dimensional
Project-Based Approach
Students conduct investigations and engage in the Engineering Design Cycle as they iteratively work towards completing the Chapter Challenge.
Students Learn Like Scientists and Engineers
Students develop important
21st-Century skills as they work collaboratively in groups and engage in scientific discourse.
Total Support
for Teachers
Professional Learning is provided by our team of education specialists.
Need more info to decide if this the right curriculum for your district or school?
Active Physical Science Curriculum Details
Active Physical Science content is based on the research on how students learn, encapsulated in the 7E Instructional Model (Elicit, Engage, Explore, Explain, Elaborate, Extend, Evaluate).
-
Chapter Challenges
Interesting and meaningful Chapter Challenges motivate students to learn and remember the science content.
-
Crosscutting Concepts
Science and engineering practices, crosscutting concepts, and core ideas are seamlessly integrated throughout the Active Physical Science curriculum.
-
Customizable Content
Follow the unit sequence listed below, or select the chapters you'd like to create your custom physical science course, available in print chapter books and interactive digital units.
Active Physical Science Research-Based Design
Active Physical Science is research-based.
Active Physical Science content was supported through National Science Foundation funding and consequently produced through rigorous, iterative, research-based development cycles. It is based on the latest research from the cognitive sciences on how students learn.
Active Physical Science students develop communication and collaboration skills.
Students develop a community of practice and a culture of collaboration, creativity, critical thinking, and communication.
The presentations of the Chapter Challenges provide students with opportunities to engage in scientific arguments using evidence and science knowledge and promote a deeper understanding through public practice.
Active Physical Science fits your unique needs.
The content is customizable and reflects the full scope of physical science content standards for high school—those identified as the Disciplinary Core Ideas in A Framework for K-12 Science Education and those of individual states and districts.
Active Physical Science Scope & Sequence
To the right is an example unit sequence, however, units can be adapted to fit your state's unique sequence.
Contact us for help customizing your physical science content to best match your standards.
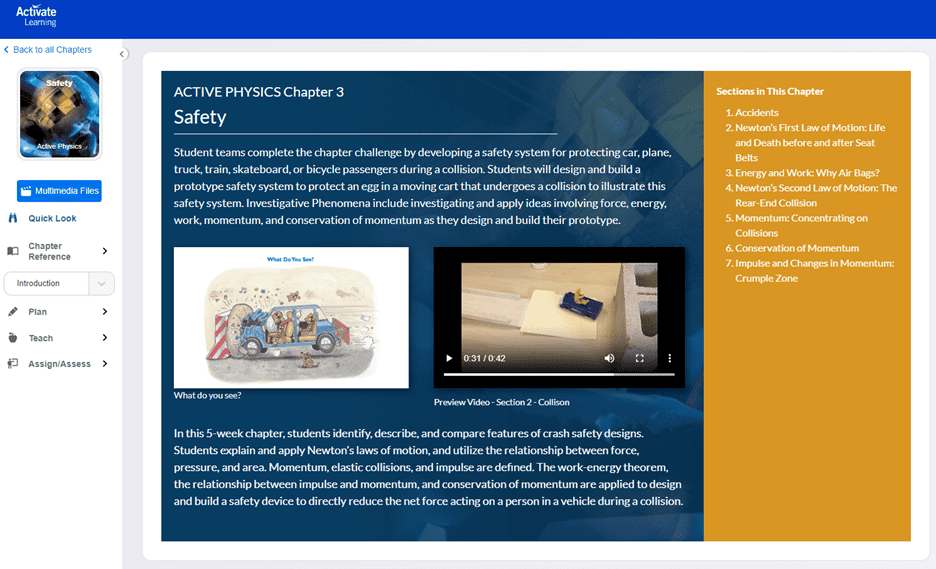
Chapter Challenge: Students demonstrate their knowledge of the physics of driving by making a presentation to a board of driving instructors.
Through a series of activities, students learn about average and instantaneous speed and then connect information about reaction time, speed, and velocity to tailgating. They explore acceleration, positive and negative, in the context of the time required to bring a vehicle to a stop and decision-making at a yellow light.
They also learn about centripetal force and acceleration and relate it to driving on curves.
Digital Platform
The Activate Learning Digital Platform (ALDP) hosts the interactive digital edition of the Active Physical Science teacher and student curriculum materials.
The platform is designed for student accessibility and inclusion and offers embedded translation for over 130 languages and text-to-speech with read-along highlighting in 35 languages.
Featuring an intuitive user experience, teachers have everything they need to Plan, Teach, Assign, and Assess lessons in a platform that is integrated with leading SIS rostering and Learning Management Systems such as Google Classroom, Schoology, and Canvas.
About The Author

Dr. Arthur Eisenkraft has taught high school physics for over 28 years. He is currently the Distinguished Professor of Science Education, Professor of Physics and Founding Director of the Center of Science and Math in Context (COSMIC) at the University of Massachusetts Boston.
Testimonials Kind Words from Administrators & Teachers.
Active Physical Science Videos
Experience Active Physical Science

Active Physical Science Principal Story

